 Novartis’ marketed drugs for neuroendocrine tumors include Afinitor ® (everolimus), which generated $1.516 billion in net sales last year, down 6% from $1.607 billion in 2015. “The isotopes don’t change the nature of the chemical entity, and they don’t change the ability for targeting.” “This approach attaches radioisotopes to therapeutic molecules to deliver therapeutics and imaging in one injection. “Theragnostics is a very straightforward proposition for physicians dealing with nuclear medicine,” AAA CEO and founder Stefano Buono told GEN in 2015. That pipeline includes prostate cancer treatment candidates 177Lu-PSMA-R2 and its companion diagnostic 68Ga-PSMA-R2 177Lu-NeoBOMB1, a new-generation antagonist bombesin analog targeting gastrin-releasing peptide receptor (GRPR)-expressing malignancies, and its companion diagnostic, 68Ga-NeoBOMB1 and Annexin V-128, a single-photon emission computed tomography (SPECT) diagnostic candidate for assessing apoptotic and necrotic processes present in a number of oncology and cardiovascular disease conditions, as well as in autoimmune disorders.ĪAA also markets two drug kits for radiolabeling somatostatin analog peptides to help diagnose somatostatin receptor-positive NET lesions, Netspot ® (formerly known as Somakit-TATE) and Somakit TOC™. “With Lutathera, we can build on this legacy by expanding the global reach of this novel, differentiated treatment approach and work to maximize AAA's broader RLT pipeline and an exciting technology platform.” “Novartis has a strong legacy in the development and commercialization of medicines for neuroendocrine tumors where significant unmet need remains for patients,” Bruno Strigini, CEO of Novartis Oncology, said in a statement. Novartis noted in its announcement today that the NETTER-1 trial demonstrated a 79% reduction in the risk of disease progression, with an interim median progression-free survival rate of 8.4 months that had yet to max out. Improve the format, traceability, uniformity, and completeness relating to clinical data-including from the Phase III NETTER-1 trial, which generated positive results. 
Provide a safety update on clinical and nonclinical studies. 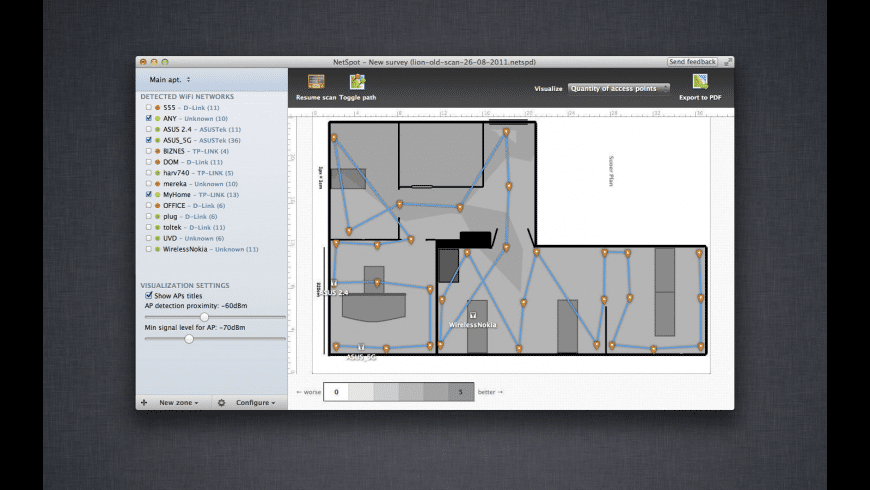
The first review in December 2016 ended with a Complete Response Letter directing AAA to:Ĭonduct subgroup analyses for gender, age, and racial subgroups, as well as other stratification factors and “important” disease characteristics. In the U.S., the FDA has set a Prescription Drug User Fee Act (PDUFA) target decision date of January 26, 2018, for Lutathera. By helping analyze every access point, NetSpot can scale up from useful-to-everyone to incredibly detailed for the true wireless networking mavens.Lutathera, which is designed to treat NETs, last month won European Commission approval for the treatment of unresectable or metastatic, progressive, well-differentiated (G1 and G2), somatostatin receptor-positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs). NetSpot offers more than 15 heatmap visualizations that help troubleshoot and manage any Wi-Fi setup. Outline your real-life Wi-Fi data on a map quickly and easily. With NetSpot, the in-depth network analysis is a breeze, thanks to its interactive color-coded heatmaps. It lets you troubleshoot and improve your network's coverage, capacity, performance, APs configurations, signal level, interference, noise, etc. NetSpot collects every detail about surrounding Wi-Fi networks and presents wireless data as an interactive table. 15+ heatmap coverage graphs are available with powerful customizable reports. You run a survey by walking, marking your position on the map, giving NetSpot a few seconds to collects data samples, watching Wi-Fi networks being detected and visualized. Wi-Fi surveys are the key feature of NetSpot. 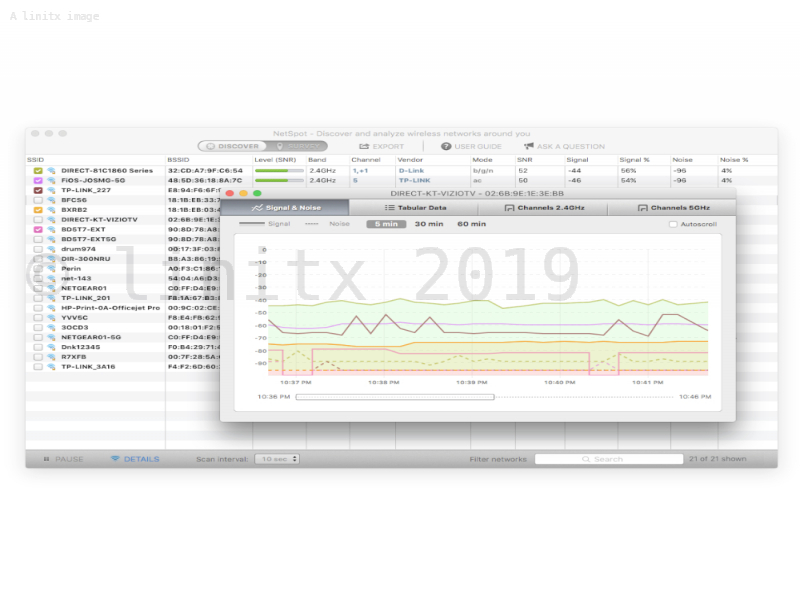
Your Wi-Fi survey app for Mac and Windows 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
